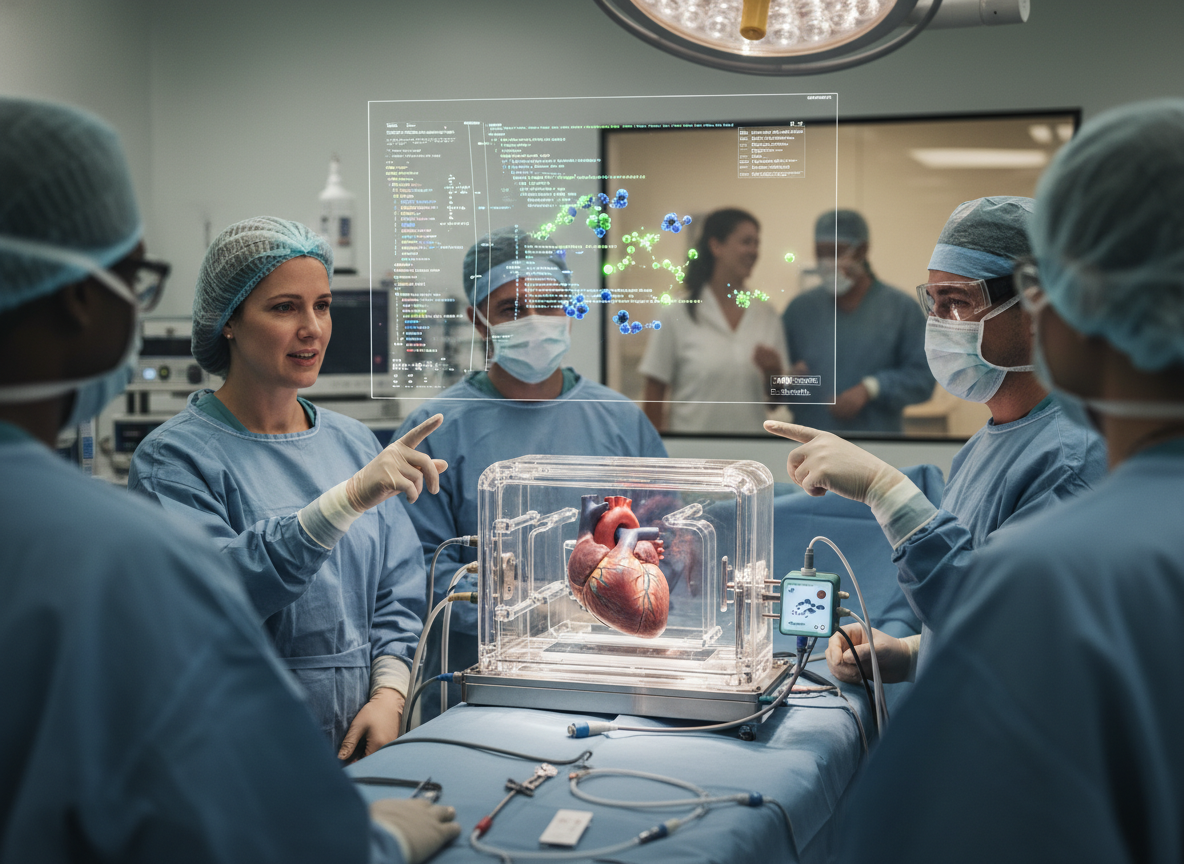
In what is being called the most significant medical advancement of the 21st century, the FDA has officially approved the 'OmniGraft' platform for clinical use. Developed by a consortium of researchers at MIT and Mass General, the platform uses CRISPR-Cas12 gene-editing technology to 'cloak' donor organs from the recipient's immune system. By neutralizing the specific proteins that trigger organ rejection, OmniGraft allows for 'universal' transplants, where any healthy organ can be successfully transplanted into any patient, regardless of blood type or HLA compatibility. This breakthrough addresses the chronic shortage of compatible organs that claims thousands of lives annually.
The approval comes after a flawless Phase III trial involving 200 patients who received 'mismatched' kidney and liver transplants. Traditionally, these patients would have required lifelong immunosuppressant drugs, which carry heavy side effects and increase the risk of infection. However, the OmniGraft-treated organs were accepted by the recipients' bodies as 'self' tissue, with zero cases of acute rejection reported over the 18-month study period. This paradigm shift in transplant medicine not only saves lives but also drastically improves the quality of life for survivors, who no longer need to worry about the constant threat of rejection.
OmniGraft works by perfusing the donor organ with a specialized 'genetic cocktail' shortly after harvest. This mixture uses viral vectors to deliver CRISPR components that permanently silence the genes responsible for presenting 'foreign' antigens on the organ's cell surfaces. The process takes approximately six hours and is performed in a specialized 'ex-vivo' machine that keeps the organ alive and functioning outside the body. This window of time allows for the genetic modifications to take hold before the transplant surgery begins, ensuring the organ is 'stealthy' by the time it enters the recipient's bloodstream.
The implications for the global organ waitlist are staggering. Currently, over 100,000 people in the United States alone are waiting for a life-saving transplant, with many dying before a match is found. With OmniGraft, the focus shifts from finding a 'match' to simply finding a 'healthy' organ. This will dramatically increase the utilization rate of donated organs, many of which are currently discarded because a compatible recipient cannot be found in time. Health officials estimate that the average wait time for a kidney transplant could drop from five years to less than six months within the next three years.
Ethical discussions surrounding the technology are already underway. While the benefits are clear, some bioethicists have raised questions about the long-term stability of the genetic edits and the potential for 'off-target' effects. However, the FDA's rigorous review process included deep-sequencing data proving that the edits were localized strictly to the donor organ and did not affect the recipient's own DNA. The agency has mandated a 10-year follow-up registry for all OmniGraft patients to monitor for any late-onset complications, a standard procedure for revolutionary gene therapies.
Financially, the OmniGraft procedure is expected to be expensive in its initial rollout, with costs estimated at $150,000 per organ treatment. However, healthcare economists argue that this is far cheaper than the cost of long-term dialysis or the multi-decade expense of immunosuppressant medications. Several major insurance providers have already announced plans to cover the procedure, recognizing the long-term savings associated with successful, rejection-free transplants. The government is also considering a 'Transplant Equity Fund' to ensure that low-income patients have access to this life-saving technology.
The success of OmniGraft is also fueling renewed interest in xenotransplantation—the use of animal organs for humans. If CRISPR can successfully cloak a human organ, it can theoretically do the same for a porcine (pig) organ that has been genetically 'humanized.' Researchers are already planning to use the OmniGraft platform in upcoming trials for pig-to-human heart transplants. If successful, this would provide an unlimited supply of organs, effectively ending the organ shortage crisis once and for all. Today's approval is the first step toward a future where no one dies waiting for a transplant.
As the first OmniGraft procedures are scheduled for later this month, the medical community is pausing to reflect on the power of genetic engineering. What was once the realm of science fiction—editing the very code of life to solve biological incompatibility—is now a standard medical tool. Dr. Anthony Chen, lead developer of the platform, stated that 'today we have finally conquered the body's fear of the other.' The legacy of April 7, 2026, will be remembered as the day we began to treat the human body as a modular, repairable system, bringing hope to millions around the globe.